Lab-Grown Meat is a Pipe Dream. We Need Lab-Grown Cheese.
Luck got us cheese, chocolate, and wine. What could we create if we really put our minds to it?
Imagine taking a time machine 20,000 years into the past, finding a prehistoric hunter-gatherer human, and serving him a plate of warm baked brie.
Imagine how completely the experience would rock his world: the richness, the mouthfeel—unlike anything he’s ever known; he’s barely got the hang of cooking meat over a fire. Imagine the look on his face as he tries to comprehend the aroma, luxurious and enticing but somehow also reminiscent of…feet?
Now imagine someone did the same thing to you.
Imagine some time-traveler came and brought you a food as gobsmacking—as earth-shattering—as the brie would be to the caveman. Something equal parts strange and wonderful: a texture that you didn’t know was possible, a new and exotic funk that even the most well-equipped flavor chemists in the world couldn't dream up.1
Spectacular new foods like that are possible. Maybe even coming soon…if we can stop chasing chickens.
Hubris
These days, a lot of the conversation around “the future of food” centers on things like lab-grown meat. Startups like Upside Foods—working toward the vision of a cruelty-free steak made from animal cells grown in a tank—have raised eye-watering sums from investors. But there’s a reason you’re not seeing it on shelves yet: without massive breakthroughs in tissue culture technology, there's practically no way for lab-grown meats to be commercially viable anytime soon.
But there is a better way. An easier way: One that's both more likely to succeed, and infinitely more rewarding once it does.
After all, even if those technological breakthroughs come tomorrow, and lab-grown meat becomes indistinguishable from the real thing…what have we really gained? Slightly cheaper chicken?2 Tissue cells grown in culture still need food, so—perhaps ironically—a lab-grown chicken nugget will probably be raised on the same old soybeans as the regular kind…once those beans have been defatted, hydrolyzed, and processed into powder.
It's still a noble goal, to be sure—the effort to transform low-quality calorie sources into tastier and more nutritious ones is the overarching theme of humanity's entire agricultural odyssey—but the approach here is too clever by half.
Think about humanity's greatest culinary achievements—the ones that literally shape the modern world: Cheese. Chocolate. Wine. Did we give up the easygoing hunter-gatherer lifestyle for grain, for crackers? No! It was only when bread—and, even better—beer came into the equation that it suddenly became a good bargain for man to put down his atlatl and pick up the plow.3
And the thing that all these foods have in common is that they are products of fermentation, which—at its heart—is the same kind of caloric upcycling that the scientists at Upside and other lab-grown meat companies are trying to pull off. But these foods don't need pressurized cleanrooms and gamma-ray-sterilized equipment. They can be made with stone-age technology, because—rather than playing God, trying to outdo nature—they’re the result of discovering nature's readymade gifts and embracing them.
Look back across history: most of the truly revolutionary advances in food science have come from people discovering how to harness the power of microbes. And looking forward, now that modern science has given us a glimpse of just how rich the microscopic world really is, it’s clear that we’ve only just scratched the surface of what’s possible.
The State of the Art
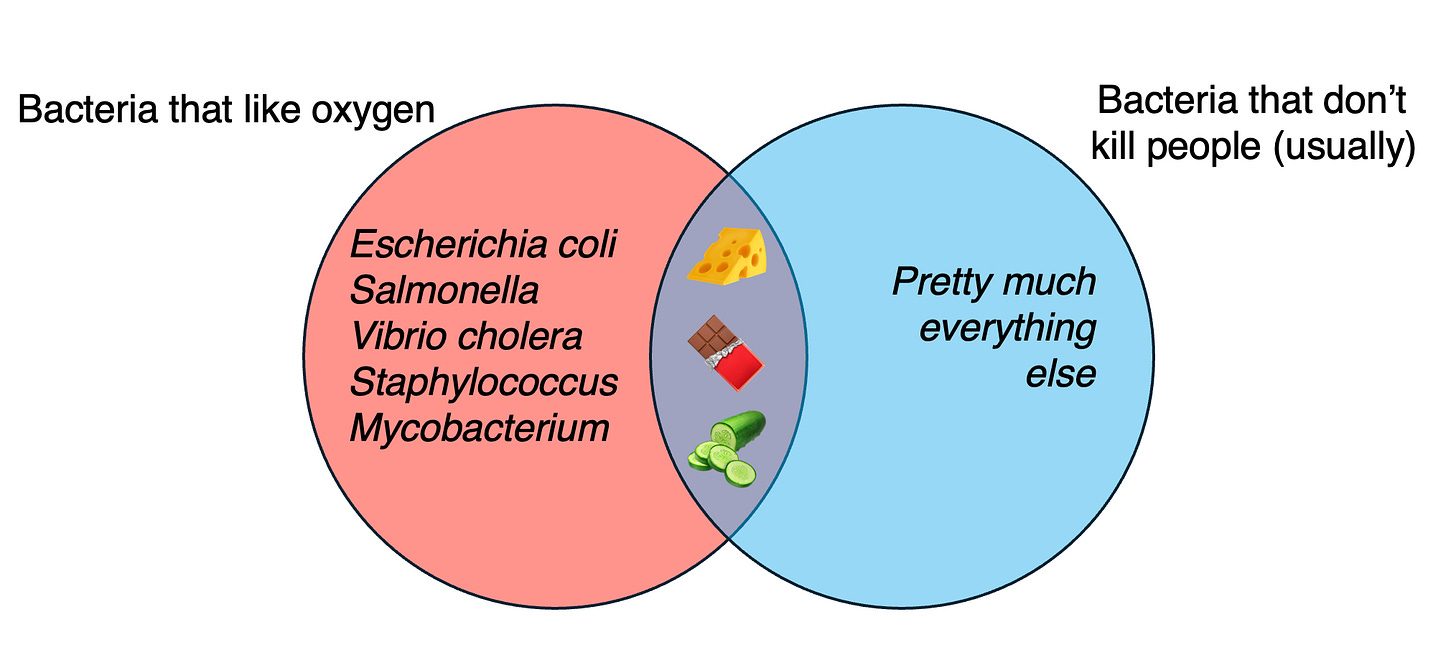
Nearly all existing fermented foods are produced by just a few different kinds of microbes. Look into the bacterial makeup of fermented foods from various cultures around the world, and you might be surprised to see the same names coming up over and over again: Japan’s famous natto, Nigeria’s Kpaye, even the coffee and cocoa beans all owe some of their unique properties to the action of Bacillus subtilis. The yeast Saccharomyces is almost singlehandedly responsible for all alcoholic beverages,4 while the various Lactobacillales give us cheese, yogurt, kimchi, pickles, sauerkraut—even prosciutto. With an assist from Propionibacterium, or Streptococcus, a Lacto can make kefir or Swiss cheese. In concert with Saccharomyces, it can make a sour beer.
But this incredible variety of products hides the fact that the organisms which help produce them represent only a select few branches on the tree of life: the useful microbes which we lucked into discovering before we even understood what they were. It’s a pretty small group, all told, because it’s limited to the set of organisms which are both oxygen-tolerant (not something you can take for granted when talking about bacteria) and non-pathogenic.
But the past century has seen revolution after revolution in biotechnology, and—while it’ll still be a long time before we can build a better chicken—we already have all the tools we need to start exploring the rest of the tree of life, to find out what kind of strange and wonderful fruits it bears. What kind of foods could we cook up with a Bacteroides or a Coprococcus as a starter culture? It’s only in the past fifty years or so that such a thing has become possible—but now, the only constraints are our imagination, and our willingness to embrace these new technologies and get creative with them.
The next new fermented food is somewhere out there. And when we find it, it’s gonna make lab-grown meat look like chopped liver.
The Problem With Eukaryotes…
Half the point of lab-grown meat lies in the potential for improvements in “feed efficiency”: how many calories of e.g. soybean you have to put into a system in order to get a tasty calorie of meat out. In a regular chicken, a lot of those calories go into producing non-tasty things, like feathers and blood and eyeballs—so if you can manage to grow just the muscle cells in a big vat, you can theoretically make big improvements.5
Unfortunately, a lot of the non-tasty bits are important for keeping the muscle cells alive.
Some of those bits are easy enough to account for with engineering: Heat the tank with electricity and you don’t need feathers for insulation. Stir it a bit, and you circulate nutrients that would ordinarily be ferried to the cells by blood. But other things aren’t so easily replaced.
Take growth factors, for example: animal cells rely on complex signaling proteins from the rest of the body to know when to grow and divide—the only kind of cells that are always switched to “grow” are cancer cells.6 Those signaling proteins are complicated, which makes them costly to produce artificially…but at least they’re possible with current tech. Other things—like the immune system—are practically irreplaceable. Because the different types of cells in our bodies are highly specialized for given functions, a vat full of muscle cells has no defenses against the germs that an ordinary animal fights off without a problem; working with animal cells in a Petri dish, a single stray cell of Mycoplasma can ruin your entire experiment. At an industrial scale, the problem is even worse: if one tank in a factory turns into a breeding ground for contamination, odds are your entire operation is toast.
We can imagine clever solutions to the problem: maybe there’s a way to keep a few immune cells in the tank along with the muscle cells, and let them patrol for any invaders. That would reduce the risk of a catastrophic failure, but it would also cut into efficiency: those immune cells need food too, and probably some weird growth factors of their own. Take this line of thinking to its logical conclusion—adding back in cell types and structures, sacrificing efficiency to improve reliability and ease of use—and before long you’ll find that you’ve reinvented the whole chicken.
Having hyper-specialized cells like this is the hallmark of eukaryotic life. It’s key to our ability to build complex and wonderful structures like brains and bones. But that hyper-specialization means that the individual cells—and especially things like muscle cells—are passive, fragile, and needy on their own. None of it works without the rest: the muscle cells expect the GI tract to be there, to break down food into individual amino acids; the circulatory system to carry those nutrients to them, fat cells to keep them at the right temperature. Take an individual cell out of that context for even a few minutes, and you have to do a lot of work to keep it alive.
All this makes your average bacterial cell look like Rambo, in comparison. Where you and I have a thousand hyper-specialized co-dependent cell types in our bodies, bacteria are scrappy generalists: every cell is a whole organism, an individual, ready to fend for itself. This is what is meant by “single-celled organism”—and it’s a powerful paradigm.
Rather than relying on an immune system to protect them from invaders, they produce acids, or alcohols, or antibiotics in order to make the environment inhospitable to their competitors—and this is part of why fermentation has been a key part of food preservation for thousands of years.
Think about blue cheese, for christ’s sake! You take this hunk of protein, carbs, and fat, leave it in a dank cave for a few months, and it comes out better than when it went in! “Oh, looks like it made a friend in there!”
Bacteria don’t need any coaxing by special growth factors in order to continue growing and dividing; as long as there’s more food and more room, they’ll keep it up. And if they’re running out of either, half of ‘em can just turn on their flagella and motor off in search of greener pastures. They’re not picky eaters, either—purified amino acids? Pah! Just throw some ground beef into the test tube, they love the stuff.

They’re hardy little guys, too. Just last week, I plated a sample onto a petri dish, and accidentally forgot to put it into the incubator before I left for the night—so it sat at room temperature, when it was supposed to be at 98°F. Animal cells left alone, thirty degrees below their preferred temperature for 14 hours? Forget it—they’re toast, they’re trash. But the next morning, I popped the plate into the incubator, and the day after that, we had growth. This is the beauty of each cell being its own organism, as self-sufficient as possible: they simply don’t know the meaning of “throwing in the towel”.
Of course, for all their virtues, this is still an apples-to-oranges comparison. At the end of the day, no bacterial byproduct is likely to make a convincing meat substitute….but who cares? What if—rather than struggling in vain to make imitation meat—we set out in search of something even better? We should be applying modern technology as creatively and experimentally as we can…rather than fixing our sights on something as boring as “meat, but from a vat”.
In many ways, the effort to make lab-grown meat a reality is an effort to get animal cells to grow and behave like microbial cells would—and at present, this takes an insane amount of effort. At a certain point, you have to ask: Is it worth it, to take over the job of extracting the protein from a soybean, hydrolyzing it into individual amino acids, then purifying and sterilizing those and stirring them into the vat, so that you can grow some chicken cells—when the chicken would do a pretty good job of all that, if you just tossed a few handfuls of beans over in the general direction of the coop?
And yes, if you go to all that trouble you can enjoy a chicken nugget without taking a life, but if we really want to wean people off of animal-based agriculture—whatever your motivation for wanting that—we need to present them with better alternatives. Hell, tofu isn’t even very good, but whoever invented it has probably done more for that cause than PETA ever could.
Fortunately, we have a frankly unbelievable toolbox to work with.
The Nose Knows
Taste and smell speak to us at a biological level. For instance: Check the ingredients on a bag of octopus-flavored potato chips from Taiwan, and you'll see "5'-ribonucleotides". Why? Because RNA is fucking delicious. Your body uses a lot of it, so your taste buds have evolved to be very sensitive to it—and those sensors are wired straight into your reward and satiation centers. The body knows what it needs, which is why the most irresistible foods are usually loaded with calories, salt, and other things that keep us going.
So if we want to find our starter for Cheese 2.0—i.e. microbes that are likely to be both safe to eat and delicious (because they possess beneficial and nutritious chemistry)—we need look no further than the human gut. It might sound gross to suggest using a bacterium isolated from poop to make a new fermented food...but only until you've smelled the result.
I've worked with a lot of different gut bacteria in isolated cultures. Some of them smell as foul as you'd expect from an organism isolated from shit...some even worse. Once in a while, though, I've uncapped a test tube to dump its contents, caught a whiff…and been absolutely entranced.
Is that...pickles? tuna salad? Neither is quite right; there was no fishiness, just a pure and enticing umami aroma with a tangy, fruity undertone. Some blend of short-chain fatty acids along with other, more obscure volatiles. It's never been more than a passing intrusive thought, because there are certain things you can’t do in a BSL2 lab—but my body was very clearly screaming drink it, DRINK IT, that’s the GOOD stuff!, and I have a feeling it was right.7
There are thousands of species in the healthy human gut: a practically infinite number of strains with unique and interesting chemistries out there, waiting to be found. Practically nobody seems to be looking. But a few weeks ago, on a hunch, I inoculated a strain of Coprococcus eutactus into some coffee that I had souped-up with a little nutritional yeast and collagen. After a few days in the incubator, it came out enhanced with an unmistakably buttery note, thanks to a little molecule called acetoin that Coprococcus makes. That’s how easy it can be—the definition of low-hanging fruit!8
Could have been a Trappist
In one sense, calling lab-grown chicken "Franken-meat" is a cheap shot. It's not green, it probably won't kill you, and I’m not holding my breath for a tap routine with Gene Wilder. But Frankenstein undeniably is the story of a man trying to create life: going to unnatural extremes to acquire the power which biology holds for herself—and finding that his best efforts can produce only a monstrosity. It reminds me of the story of a medieval monk, who spent practically his whole life walled up in the monastery tinkering with gears and cogs and springs, and after forty years unveiled his creation: a clockwork monk, a few feet high, that could walk and kneel and offer silent prayers. A marvel for its time…but the first time I heard that story, I thought “Man, you could have just had a kid”.
That’s the foreseeable future with lab-grown meat. Not a monstrosity, and maybe even a technical marvel—but a sad shadow of the real thing. Because if you zoom way, way the fuck in, the real-live monk is a clockwork monk. Collagen coilsprings give your skin its bounce. In your mitochondria, a proton-gradient breeze turns the windmill blades of ATP synthase, clicking a phosphorous into place, charging your batteries. Protein strings fold into sheets, which fold into little origami machines, which produce more protein strings. It’s all physical, just optimized to a scale we can barely fathom—and in our efforts to imitate it, we’re only a little better off than the fumbling monk.
But why try to imitate nature, when we can innovate with it? “Meat 2.0” is ambitious, sure, but so was Dr. Frankenstein—who might have had a lot more luck creating life, if he’d simply set his sights a little lower and found a nice girl to settle down and have kids with. After all, at the end of the day, which monk brought more delight and wonder to the world: the one toiling away on his little toy man, or the lucky Trappist who discovered the miraculous funk of a Belgian tripel?
—🖖🏻💩
Most of this piece was written over a year ago, as part of the Ideas Matter fellowship, sponsored by Asimov Press. Many thanks to Niko McCarty and the whole team at Asimov for their efforts, and many apologies for letting this sit in drafts until the publication was defunct. If you liked this piece, please click the button below to subscribe for free so that future posts come straight to your email inbox.
Largely because, if the Funk is strong in you, you don’t go into organic chemistry—or if you do, you end up like Shulgin.
And a slightly cleaner conscience, if that sort of thing bothered you before.
The oldest known book in the world, the Epic of Gilgamesh, begins with the story of a wild man named Enkidu—a beastly, hairy creature who drinks from a watering hole with the other animals. Gilgamesh arranges for him to try some beer (and, full disclosure, get laid) after which the other animals shun Enkidu; he's no longer one of them. Literary types will tell you that Enkidu's tale is a parable representing man's primal past, the transition to civilization facilitated by agriculture, yada yada yada…but I don’t know if they had invented symbolism in literature yet, and it seems equally likely to me that some version of these events just actually happened—where seven beers and some early-Mesopotamian pussy was all it took to convince a holdout that this whole “civilization” thing was worth a shot. Art imitates life, y'know.
Central America has an unusual drink called pulque, made from agave sap fermented into alcohol by Zymomonas mobilis—one of the few alcoholic drinks produced by a bacterium. Always wanted to try some; I hear it’s pretty gross.
Although the feed efficiency for a modern broiler is something like 0.3—meaning: ten calories in, three calories of meat out. That’s hard to beat.
I suspect that, at every company doing lab-grown meat, the idea has been hesitantly floated by R&D before being shot down by Regulatory and Marketing at faster-than-lightspeed. “Well, if we used cancer cells—”
“—You want to make cancer nuggets, Josh? Don’t answer that, I don’t care; we’re not doing it.”
I don’t make a regular practice of inhaling from test tubes of live bacteria, but I started making casual notes on which strains smelled delicious, or funky in an appealing way when I happened to notice one. Curiously enough, some of these “sniff picks” ended up at the top of the list in our assays for things like anti-inflammatory activity.
Funnily enough, Coprococcus is Latin for “poop berry”—a reference to the bacterium’s appearance under the microscope: clusters of spheroidal cells, almost like grapes. Perhaps it’s only natural, then, that the dingleberry represents such low-hanging fruit.


IMO it is the partnership of microbial fermentation with feedstocks which are easy to grow but not considered especially edible is the real low hanging fruit. Lots of weedy semi-edible plants which are one complementary detoxification enzyme away from being delicious and dependable.
As an example, we have a hyper productive legume tree (Castanospermum). The local foragers used to roast and slice the giant starchy seeds, then soak them in running water for a week to leech the nasty alkaloid. The right bacteria would eat that molecule with a spoon and short cut the lengthy process (though the saponins could be more of a challenge).
Ahh, the dingle-est of berries...and cancer-nuggets!
And this set of systems we call a body...almost seems like it was designed? (spoiler alert, it was :)
I can't look at these molecular machines and the complex iterative dance that keeps us alive and think it was change by chance that developed it.
RNA? Where there is a code, there is a coder.
Cheers...a thought provoking piece for us humans that will eat almost anything.